Atomic mass number of calcium12/22/2023  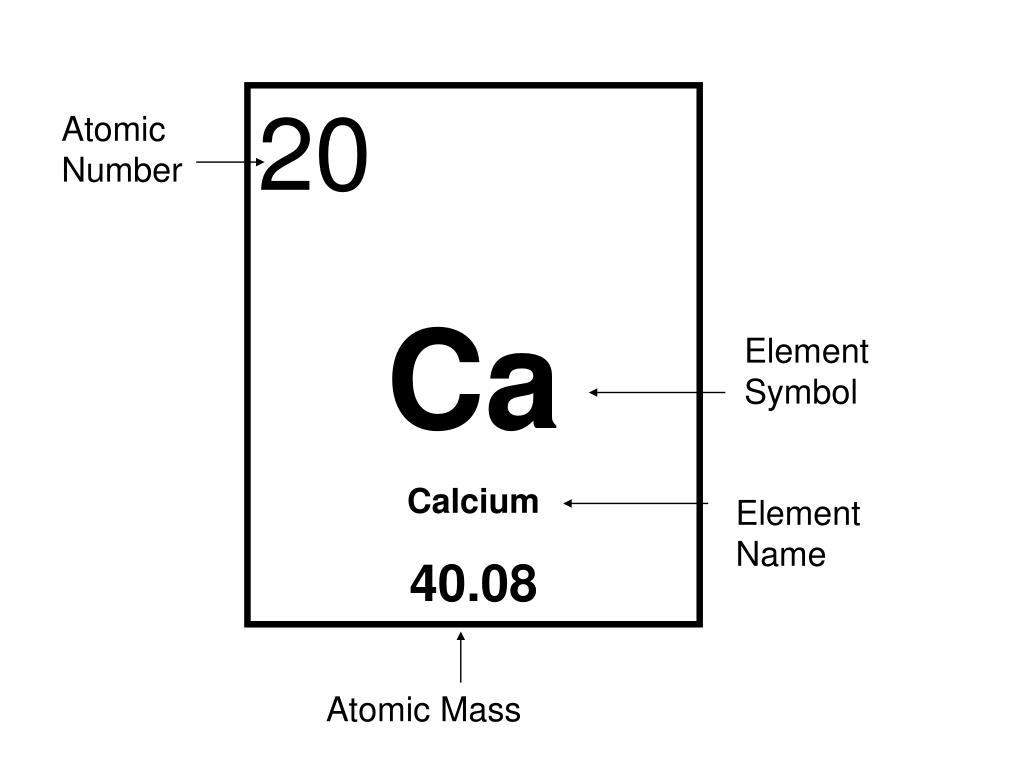
Table with the Atomic Masses of Calcium Isotopes In the case of calcium, this results in an average atomic mass of about 40,078 u. The calculation of atomic mass is done by multiplying the mass of each isotope by its relative abundance and adding the results. Other isotopes of calcium, such as Ca-42 and Ca-48, are less abundant and have a minor effect on the average atomic mass. Additionally, calcium-44 (Ca-44) is another common isotope, making up approximately 2.1% of natural calcium. The atomic mass of calcium is calculated by taking into account the relative abundance of its natural isotopes.Ĭalcium has several isotopes, the most abundant being calcium-40 (Ca-40), which represents approximately 96% of all natural calcium. Calculation of the Atomic Mass of Calcium The atomic mass of calcium is a physical and chemical concept that refers to the average mass of all calcium isotopes found in nature, weighted by their relative abundance.Ītomic mass is expressed in unified atomic mass units (uma) or daltons (Da), and for calcium, its average atomic mass is approximately 40,078 u. However, the relevance of calcium goes beyond human biology and extends to a wide range of scientific fields. The minimum intake is about 4.7 mg in average.Calcium, a chemical element essential for life, is known for its role in the health of bones and teeth. Humans take several forms of potassium but the most common one is potassium chloride. Problem 1: Which is the most common form of potassium taken by us? 
It can also lead to dental changes, alterations in the brain, etc Lack of calcium in the blood can cause a disease called “Hypocalcemia” also known as calcium deficiency in the human body.We can find 2% of the calcium in the human body. Calcium is important for both animal and plant life.Calcium hydroxyl phosphate is the principal inorganic element of teeth and bones.Calcium carbide is used for the production of plastics and acetylene gas.Calcium concatenate is used as a food additive.It is also used as an alloying agent for the production of some metals.It can be used as a reducing agent in the metal extraction process.It is needed for nerves to carry messages between the brain and every body part.Calcium helps to maintain strong bones to perform many necessary functions. .png)
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
